For Professional Use Only
The Intelligent Fingerprinting VSS-GP COVID-19 Saliva Test makes testing far easier for all ages:
- Saliva-based – for simpler and quicker collection of samples
- Detects SARS-CoV-2 antigen, including the Omicron variant
- Reduced plastic waste – thanks to more environmentally-friendly test strip design
- Designed to support COVID-19 testing in schools, workplaces, universities, care homes, healthcare and public events
- Entirely UK researched, developed and manufactured
- CE marked for professional use
Available to order, subject to regulatory approval in your country. For more information call +44 (0)1223 941941.

The Intelligent Fingerprinting VSS-GP COVID-19 Saliva Test uses lateral flow technology to detect the SARS-CoV-2 antigen, including the Omicron variant, using a saliva sample.
Unlike existing COVID-19 tests that require unpleasant, invasive nose and throat swabs, our test uses a simple swab to collect a saliva sample just from the inside of the cheek.
The test strip is deliberately simple in design and engineered to keep plastic usage to a minimum – with none of the plastic casing that is usually associated with lateral flow tests.
Because it is so easy to collect a saliva sample, the test is particularly suitable for use with more vulnerable people and children. It is CE marked for professional use, facilitating frequent testing across a range of applications including schools, workplaces, public events, care homes, healthcare, airports and prisons.
Test results are available in 20 minutes.

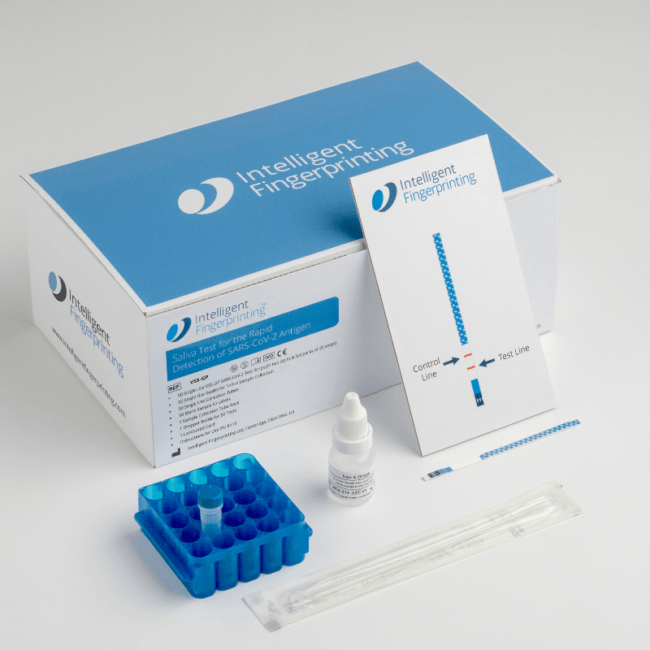
The swab is used to collect the saliva sample from the inside of the lower cheek, which is then mixed with a reagent buffer that has been pre-added to the collection tube in order to release the virus. The screening test is performed by inserting the tip of the Intelligent Fingerprinting VSS-GP test strip into the sample, providing a positive or negative result at 20 minutes. No instrumentation is required.
Our test uses gold immunonanoparticle technology to enable the visual detection of bound antigen via a control line and a red line on the test strip when a saliva sample is positive for the SARS-CoV-2 antigen.
The test comprises a four-part kit with 50 single-use COPAN CE-marked swabs for saliva collection, 50 single-use plastic collection tubes with lids, and 50 single-use VSS-GP SARS-CoV2 test strips, along with four dropper bottles containing the reagent buffer.

Clinical precision testing of Intelligent Fingerprinting’s VSS-GP COVID-19 Saliva Test has resulted in 100% sensitivity and specificity (n = 400 results) using RT-PCR verified samples containing SARS-CoV-2 virus (lowest viral titre Ct 28.5).
The SARS-CoV-2 monoclonal antibody we use for antigen detection has been tested against the Omicron Nucleocapsid Protein and the VSS-GP COVID-19 Saliva Test has strong recognition of this variant.
This product is CE marked for professional use under the In Vitro Diagnostics Directive.
Available To Order Now
Call +44 (0)1223 941941
Manufacturing capacity at our Cambridge plant, utilising existing equipment and ISO 13485 quality control, is ready to support the production of millions of tests per month, and we also anticipate we will be working with existing and new distribution partners to make this test available across multiple markets.
For further information about products or partnership opportunities, please contact us.

